Webinar: Experten-Interview - Digitalisierung der Produktionsauftragsakte
Medical technology digitization from the market leader: security, compliance, and state-of-the-art AI technology
Digitalization in medical technology is the key to the future success of your company. It creates clarity, security, and scalability for the further development of your business. With Microsoft Dynamics 365 and solutions from COSMO CONSULT, you can connect all areas of your company from product development, manufacturing, quality assurance, and management to delivery and service in a continuous end-to-end process flow.
From now on audit trails, e-signatures, and UDI management are a breeze with traceability at the touch of a button, approvals in minutes, and real-time transparency through AI-supported analyses. The Microsoft Cloud platform, modular implementation, and integrations with industry-standard partner solutions (such as NiceLabel) and central MedTech databases (such as EUDAMED/GUDID) ensure scalability and investment protection. Rely on a future-proof IT architecture and proven confirmation of your product quality.
Challenges in medical technology – how we tackle them
Fact check: COSMO's contribution to medical technology digitization in figures
50+
experts for regulated industries (MDR, ISO 13485, GAMP 5)
30+
successful projects in medical technology
10+
years of experience in the MedTech industry
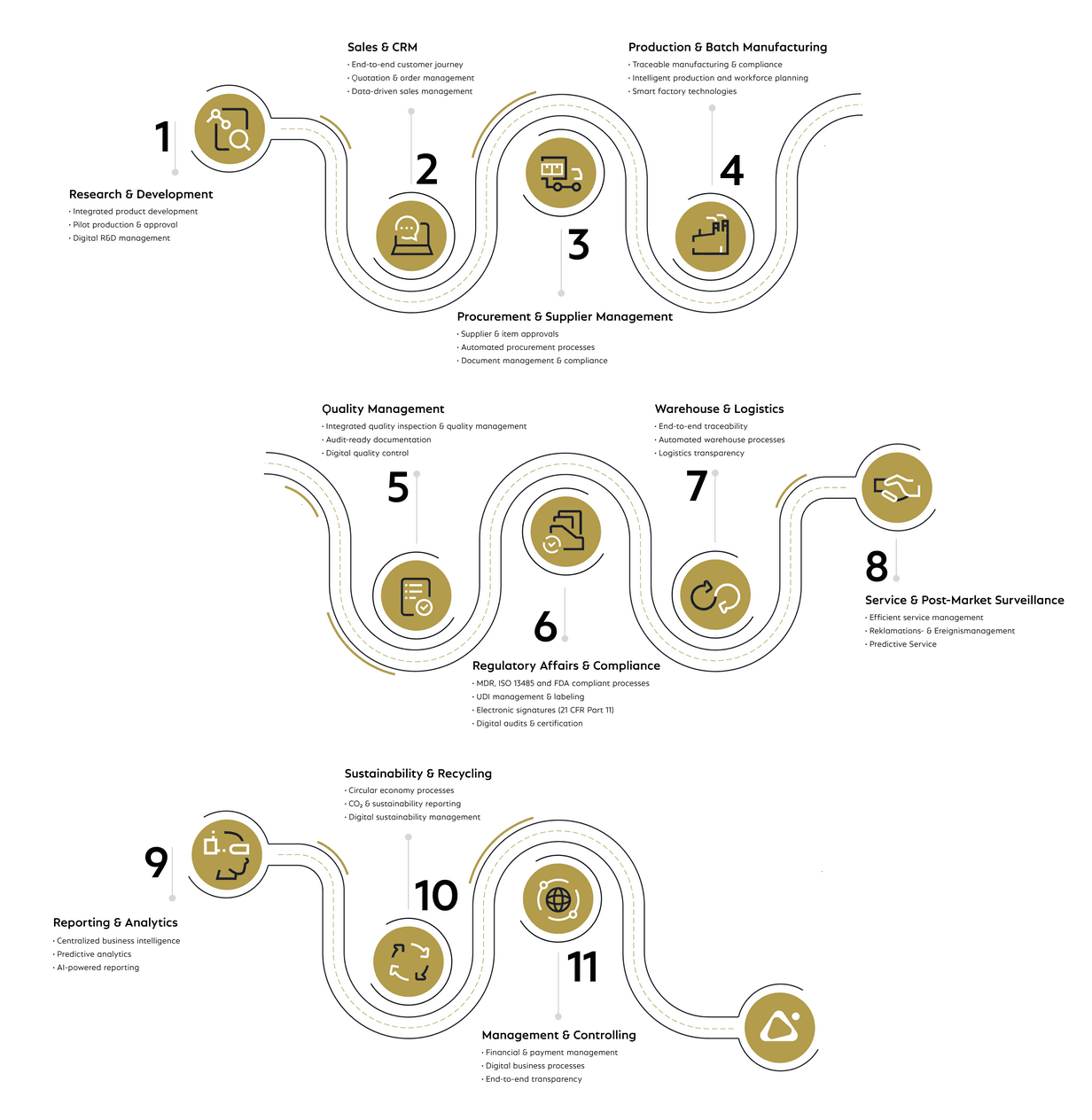
How COSMO CONSULT supports MedTech companies across the entire value chain

AI in medical technology: From analysis to process optimization
The future is now: Artificial intelligence is revolutionizing the entire MedTech industry. With Microsoft Dynamics 365 and COSMO CONSULT, you can now use the world's leading technology for AI-supported analysis in quality, planning, and service. With anomaly and pattern recognition, you can make a quantum leap in predictive scenarios, reduce waste, and increase process reliability.
The potential is virtually limitless: AI helps you make informed decisions in real time based on your company's collective knowledge. And it opens up new opportunities for innovative ideas and the further development of your business by automating routine tasks. This allows you to secure your innovative edge and future viability – while meeting the highest compliance requirements.
Quality management at the highest level: digital and fully integrated
Consistent quality processes
With COSMO CONSULT and Microsoft Dynamics 365, you can centrally control your quality management, CAPA, and audit trail across all areas of your company. Digital workflows and e-signatures speed up approvals and ensure complete traceability.
Supplier evaluation & complaint management
Integrated supplier evaluation and digital complaint processes improve your data quality and reduce error rates. This enables you to ensure consistent quality and efficiently meet regulatory requirements.
Risk management and compliance in medical technology
Regulatory compliance is a must-have in the MedTech industry. COSMO CONSULT solutions support MDR, FDA, and ISO 13485, so you are equipped to meet all requirements. Audit trails and e-signatures ensure systematic traceability. And with COSMO Computer Software Assurance (CSA), you can significantly reduce your validation efforts.
Other built-in features: FDA/EU-compliant UDI management with NiceLabel integration, support for CAPA and deviations, and easy data integration through standard interfaces to MES, PLM/PDM, and DMS.
Cloud ERP and data security in medical technology
Scalable cloud architectures
World-leading cloud technology: With Microsoft Dynamics 365 and Microsoft Cloud, you benefit from an easily scalable cloud platform that includes regular updates, security by design, and international rollout capability. Your IT costs decrease while innovation cycles accelerate.
Data security & compliance
COSMO CONSULT ensures that your cloud platform operates in compliance with GDPR and provides maximum security through a role-based access concept and the isolation of your company data. Automated updates and validation according to GAMP 5 ensure that your compliance requirements are always met – for maximum security and investment protection.
What we’re proud of: Medical technology companies that rely on us
Current awards
Frequently Asked Questions
Found what you were looking for?
Start your intelligent search now