Product of the month - cc|quality management
In many industries, quality is the decisive selling point. A professional quality management system supports companies in the process of adding value and is closely linked with their operational business processes. This works best if the ERP system also has quality management capabilities. Microsoft Dynamics 365 (formerly Microsoft Dynamics NAV and Navision) can be extended with the add-on software cc|quality management to provide these capabilities.
In sectors such as health care, pharmaceuticals, the food industry, and aerospace nothing is done without quality assurance in place. In these fields, end-to-end, professional quality management is required by law, especially on account of the potential risks to the population. But quality is very important in other industries, as well. The reason is obvious: negligence can be very expensive for companies. Not only because of the threat of fines and claims for damages, but also because poor quality undermines a company’s competitive position. Good quality management helps:
- To avoid quality defects and deviations from the standard in advance
- To detect and analyze errors faster
- To identify, track, and recall defective items
- To make test and manufacturing processes uniform, as well as
- To meet customer requirements and quality standards throughout the company.
Quality as part of business processes
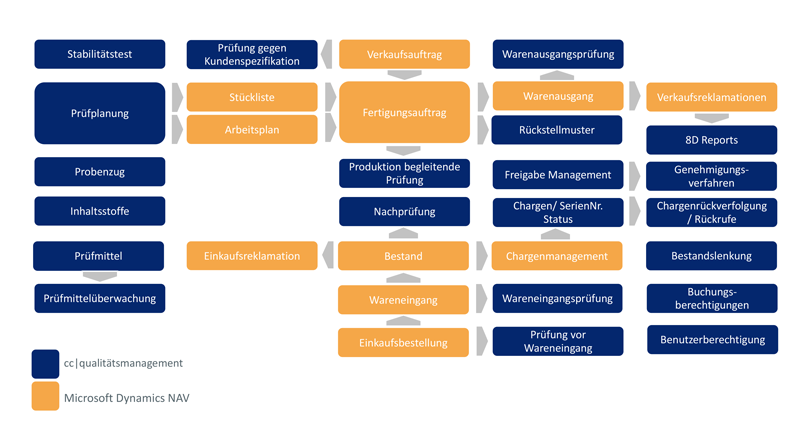
Modern quality management is an integral part of all phases of operational value creation - from the arrival of incoming goods, through production, to the delivery of the finished product to the customer. "Quality" thus becomes a key topic, affecting all parts of the company equally. So what could be more obvious than to link quality management functions tightly with the operative business processes on the software side? This works best when it is possible to extend the ERP system with additional software for quality management. One such solution is cc|quality management for Microsoft Dynamics 365.
Adaptable to operational needs
If products or materials deviate from previously defined specifications, it is first necessary to determine what the problem is. At what point are the deviations being produced? How can the affected materials, parts or articles be identified reliably? cc|quality management documents the entire life cycle of your products - from the arrival of incoming goods to use by the customer. Exactly where in the value chain quality checks are conducted is up to you. You can also create reporting processes and approval procedures in case of quality defects as required by legal regulations or operational procedures.
Finding defective goods faster
Every new delivery of goods is given a batch or serial number when it arrives. The number can be used as a unique identifier and to track the materials or parts throughout their entire lifecycle. Every single step and every change in the context of operational value creation is seamlessly recorded in the history list. If there are deviations in quality, the affected batch or lot can be found quickly. Your employees are thus in a position to answer customer and government inquiries within a very short period of time or to recall goods immediately. If the batch is of limited quality but is still usable, status labels such as "quarantine" or "restricted use" can help to sort out defective stock from regular stock. This makes it unnecessary to inconveniently store the defective stock in a separate area. In sectors such as the pharmaceutical and food industry, expiration dates also play a special role. Here, cc|quality management supports your employees through a transparent shelf life management system that gives you convenient control of expired goods.